|
These bonds exist along with the sigma bond with the restricted rotation. Sigma bonds exist independently and they allow the free rotation. In its bond formation, overlapping orbitals should be two un-hybridized orbitals. Also, it can be two pure orbitals and two hybrid orbitals. The important difference between the sigma and pi bond Sr No.ĭuring its formation, overlapping orbitals can be one hybrid orbital and a single pure orbital. Pi bond shows the properties of conjugated systems because all such systems have the alternate single and double or triple bonds. Pi bonds are loosely bounded and hence these are weak bonds. In these bonds, one or more electron pairs can move from one other atom and hence making the delocalized chemical bonding. 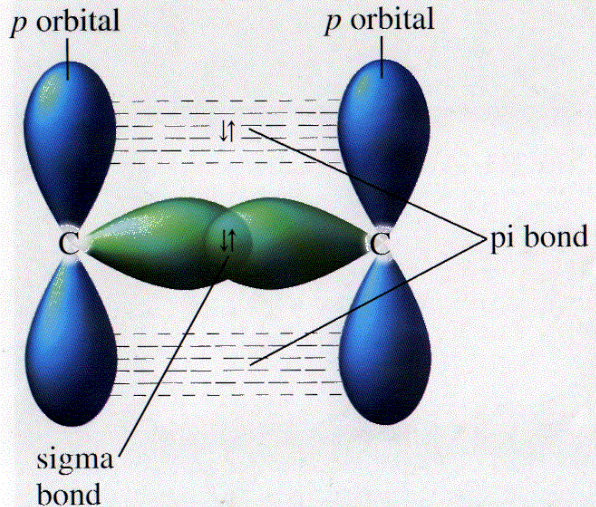
Pi bond participates in the resonance without showing hyper-conjugation. Its stability is less due to higher energy. This bond is formed by sidewise or parallel overlapping between two orbitals such as py-py orbitals or Pz-Pz orbitals. Also, their many properties like bond lengths, bond energies, and dipole moments can be predicted easily. Compounds with localized sigma bonding can be completely elaborated by a single Lewis structure. Therefore sigma bond is stronger than other bonds. 
In these bonds, electron pairs exist between two atoms with the localized bonding. They show no bond resonance or hyper-conjugation. These bonds are more stable with their lower energy. This may be between two ‘s’ orbitals, two ‘p’ orbitals, or even in between one ‘s’ orbital and one ‘p’ orbital. Sigma bond is formed by the overlapping between two atomic orbitals in face to face. 
The bond formed by the face to face overlapping between two atomic orbital’s ( between s – s orbital’s, p – p orbital’s or s – p orbital’s ), is called a sigma bond. 3.1.1 FAQs about sigma and pi bonds: What is Sigma Bond?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
